Chapter 9
Biomass Energy
KEY CONCEPTS
- Biomass production
- Composition and properties of producer gas
- Theory and process of gasification
- Gasifier types and comparison
- Use of a biomass gasifier
- Biomass feed (fuel)
Solar energy by means of photosynthesis stores energy in trees and plants that can be converted into liquid fuels suitable for internal combustion engines. Similarly, ethanol could be produced from cellulose on large scale. There is no doubt that rising energy costs will lead to more concentrated research of such biological system; such that, energy gains made via plant photosynthesis using intensive systems are subsequently more than that lost in the conversion of biomass energy content into storable high energy fuels (i.e., ethanol or methane). The growth of sugarcane and its fermentation to ethanol may be considered to be the most favourable for the marginal net energy production process, which is suitable for Indian climatic conditions. Biomass is used for heating, electric power generation, and combined heat and power. Several methods are used for the conversion of biomass into useful energy, such as electricity generation by direct burning of biomass, synthesis gas production by gasification, and methane gas production by anaerobic digestion. However, following issues may be thoroughly investigated for implementing biomass production scheme:
- The types of vegetation best suited for an intensive energy plantation and bio generation selection criteria.
- The type and availability of land for growing energy crops and plant material productions.
- Harvesting for conceptual plantation.
- Techno-economic comparison of firing crops directly for electric power generation with conversion to clean fuel gas (methane or low BTU gas) either at the farm site or at the selected market.
9.1 BIOMASS PRODUCTION
Sun is the primary source of all kinds of available raw energy resources including biomass. The sunlight energy is transferred to biosphere by the photosynthesis process that occurs in plants, algae, and some types of bacteria.
Plant matter created by the process of photosynthesis is called biomass. Photosynthesis is a natural radiation. In its simplest form, the final reaction of this process can be represented as follows:
![]()
It is seen that in the process, water and carbon dioxide are converted into organic material.
The term biomass refers to those organic matters that are stored in plant and trees in the form of carbohydrate (sugar). It is then transferred through food chains in humans, animals, and other living creatures and their wastes.
The term biomass includes all plant life: trees, agricultural plants, bush, grass and algae, and their residues after processing. Biomass may be obtained from forests woods, agricultural lands, arid lands, and even waste lands. It may be obtained in a planned or unplanned manner. The term is also generally understood to include animal and human waste.
Biomass has the advantage of controllability and availability when compared to many other renewable energy options. There are a variety of ways of obtaining energy from biomass. These may be broadly classified as direct methods and indirect methods.
9.1.1 Direct Methods
Raw materials that can be used to produce biomass energy are available throughout the world in the following forms:
- Forest wood and wastes
- Agricultural crops and residues
- Residential food wastes
- Industrial wastes
- Human and animal wastes
- Energy crops
Properly managed forests will always have more trees, and agricultural and energy crops management will always have crops; further, the residual biological matter are taken from those crops.
Raw biomass has a low energy density based on their physical forms and moisture contents and their direct use are burning them to produce heat for cooking. The twin problems of traditional biomass use for cooking and heating are the energy inefficiency and excessive pollution.
Inefficient way of direct cooking applications, inconvenient and inefficient methods of raw biomass transportation and storage and high environmental pollution problems made them unsuitable for efficient and effective use. This necessitated some kind of pre-processing and conversion technology for enhancing the usefulness of biomass.
9.1.2 Indirect Methods
Biomass can also be used indirectly by converting it either into electricity and heat or into a convenient usable fuel in solid, liquid, or gaseous form. The efficient conversion processes are as follows:
- Thermo-electrical conversion: The direct combustion of biomass material in the boiler produces steam that is used either to drive a turbine coupled with an electrical generator to produce electricity or to provide heat for residential and industrial system. However, the boiler equipment are very expensive and energy recovery is low. Fortunately, improved pollution controls and combustion engineering have advanced to the point that any emissions from burning biomass in industrial facilities are generally less when compared to the emissions produced when using fossil fuels (coal, natural gas, and oil).
- Biomass conversion to fuel: Under present conditions, economic factors seem to provide the strongest argument of considering biomass conversion to fuel such as fermentation and gasification. In many situations, where the price of petroleum fuels is high or where supplies are unreliable, the biomass gasification can provide an economically viable system, provided the suitable biomass feedstock is easily available. Biomass conversion processes can be classified under two main types:
- Thermo-chemical conversion includes processes such as destructive distillation, pyrolysis, and gasification.
- Biological conversion includes processes such as fermentation and anaerobic digestion.
Gasification produces a synthesis gas with usable energy content by heating the biomass with less oxygen than needed for complete combustion. Pyrolysis yields bio-oil by rapidly heating the biomass in the absence of oxygen. Anaerobic digestion produces a renewable natural gas (methane gas) when organic matter is decomposed by bacteria in the absence of oxygen.
As a result, it is often advantageous to convert this waste into more readily usable fuel form like producer gas. Hence, it is the attractiveness of gasification.
The efficiency of a direct combustion or biomass gasification system is influenced by a number of factors such as including biomass moisture content, combustion air distribution and amounts (excess air), operating temperature and pressure, and flue gas (exhaust) temperature.
9.2 ENERGY PLANTATION
An interesting approach for the large-scale planned use of wood is the ‘energy plantation’ approach. In this scheme, selected species of trees are planted and harvested over regular intervals of time in a phased manner so that wood is continuously available for cooking or allied purposes.
Energy plantations include, amongst others, pine, cottonwood, hybrid poplar, sweetgum, and eucalyptus. Much of the emphasis has been on hardwood plantations due to their ability to coppice, continued genetic improvement programs as well as the opportunity to combine fast growth and wood. Some important trees grown in India for this purpose are eucalyptus, babool, and casuarinas.
A rich experience of commercial energy plantations management system in varied climatic conditions has emerged during the past 4–5 decades. Improvements in soil preparation, planting, cultivation methods, species matching, biogenetics and pest, disease and fire control have led to enhanced yields.
It has been suggested that electrical power be produced by the energy plantation approach, the wood grown in this manner being used as a fuel for the boilers of a conventional power plant. The technology of biomass-based electric power plants is well established in the USA and Europe and there are over 500 such plants use wood, wood waste, and various types of agricultural waste. When a photosynthetic conversion efficiency of around 1% is assumed, it is estimated that a 1,000 MW power plant may require an area of about 1,000 km2 for the energy plantation. Although this is a large area, it should not be difficult to provide in most countries since the land required need not displace agricultural land. However, care has been taken so that there is no danger of monoculture weakening the ecological system.
9.3 BIOMASS GASIFICATION
Biomass gasification is a process of partial combustion in which solid biomass usually in the form of pieces of wood or agricultural residue is converted into a combustible gas mixture.
Gasification, which is incomplete combustion of carbonaceous fuels, can be represented with the following sub-stoichiometric equation.
Biomass + air → carbon monoxide (CO) + carbon dioxide (CO2) + methane (CH4) + hydrogen (H2) + nitrogen (N2) + water vapour.
Gasification produces a synthesis gas with usable energy content is produced by gasification in which biomass is heated with less oxygen than that needed for complete combustion.
As a result, a gaseous mixture of carbon monoxide (CO), carbon dioxide (CO2), methane (CH4), hydrogen (H2), and nitrogen (N2) called producer gas is obtained.
Producer gas can be used
- to run internal combustion engines (both compression and spark ignition)
- as substitute for furnace oil in direct heat applications and
- to produce, in an economically viable way, methanol
Methanol is an extremely attractive chemical that is useful both as fuel for heat engines as well as chemical feedstock for industries. Since any biomass material can undergo gasification, this process is much more attractive than ethanol production or biogas where only selected biomass materials can produce the fuel.
Gasification processes involved with biomass are as follows:
- Drying of fuels: It is the process of drying biomass before it is fed into gasifier.
- Pyrolysis: It is a process of breaking down biomass into charcoal by applying heat to bio-mass in the absence of oxygen.
- Combustion: All the heat required for different processes of gasification are made available from combustions.
- Cracking: In this process, breaking down of large complex molecules (such as tar) takes place when heated into lighter gases.
- Reduction: Oxygen atoms are removed in this process from the combustion products (hydrocarbon) molecules and returning them to combustible form again.
9.3.1 Low Temperature Gasification
When gasification of biomass is carried out at 750°C to 1,100°C, it is referred to as low temperature gasification. The gas produced has relatively high level of hydrocarbons. It is used directly to either burn for steam production and generation of electricity or cleaned and used in internal combustion engine or combined heat power (CHP).
The producer gas is a mixture of carbon monoxide (CO), carbon dioxide (CO2), hydrogen (H2), methane (CH4), and nitrogen from air. The gas mixture composition depends on gasifiers.
9.3.2 High Temperature Gasification
It is carried out in temperature range of 1,200°C–1,600°C and gas product is referred to as synthesis gas (Syngas). It contains high proportion of CO and H2 and is convertible to high quality synthetic diesel biofuel compatible for use in diesel engines
9.3.3 Composition and Properties of Producer Gas
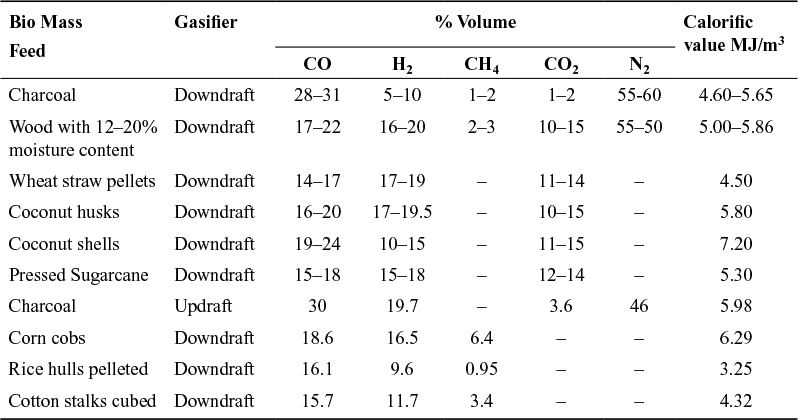
The producer gas is affected by various processes as abovementioned, and hence one can expect variations in the gas produced from various biomass sources. The composition of producer gas is highly dependent upon the inputs to the gasifier and gasifier design.
Table 9.1 lists the composition of gas produced from various sources. Nitrogen affects the maximum dilution of gas and almost 50%–60% of gas is composed of noncombustible nitrogen. The use of oxygen instead of air will be beneficial for gasification with due regards to the costs of oxygen. Nevertheless, production of a high energy quality methanol may justify the cost of oxygen.
On an average, 1 kg of biomass produces about 2.5 m3 of producer gas at S.T.P and consumes about 1.5 m3 of air for combustion. For complete combustion of wood, about 4.5 m3 of air is required.
Table 9.1 Composition of Producer Gas from Various Biomass Feed
The average energy conversion efficiency of gasifiers is defined as
 (9.2)
(9.2)
Example 9.1
One kilogram of wood produces 2.5 m3 of gas with average calorific value of 5.4 MJ/m3. Average calorific value of wood (dry) is 19.8 MJ/kg. Calculate the average conversion efficiency of wood gasifier.
Solution From Eq. (9.2), ηgs = (2.5 × 5.4 × 106) /(19.8 × 106 × 1) = 68.18%.
The average energy conversion efficiency of wood gasifiers is about 60%–70%.
9.3.4 Temperature of Gas
The average gas temperature produced by gasifier is about 300°C—400°C and it may even attain a higher temperature of approximately 500°C, if partial combustion of gas is taking place. Partial combustion of biomass can be eliminated by increasing air flow rate higher than the design value.
9.4 THEORY OF GASIFICATION
Gasification may be considered as a special case of pyrolysis where destructive decomposition of biomass (wood wastes) by heat is converted into charcoal, oils, tars, and combustible gas. It is referred to as the partial combustion of solid fuel (biomass) and takes place at temperatures of about 1,000°C. The reactor used for gasification is called a gasifier.
The complete combustion of biomass produces biomass gasses that generally contain nitrogen, water vapour, carbon dioxide, and surplus of oxygen. However, in gasification (with incomplete combustion), as shown in Figure 9.1, product gas contains gases such as carbon mono oxide (CO), hydrogen (H2), and traces of methane and non-useful products such as tar and dust. The production of these gases is obtained by the reaction of water vapour and carbon dioxide through a glowing layer of charcoal. Thus, the key to gasifier design is to create conditions such that

Figure 9.1 Products of gasifiers
- Biomass is reduced to charcoal
- Charcoal is converted at suitable temperature to produce CO and H2
Typically, the volumetric composition of biomass-based producer gas is as follows:

9.5 GASIFIER AND THEIR CLASSIFICATIONS
Biomass gasifier may be considered as a chemical reactor in which biomass goes through several complex physical and chemical processes and producer or syngas is produced and recovered.
There are two distinct types of gasifier:
- Fixed bed gasifier: In this gasifier, biomass fuels move either countercurrent or concurrent to the flow of gasification medium (steam, air, or oxygen) as the fuel is converted to fuel gas. They are relatively simple to operate and have reduced erosion.
Since there is an interaction of air or oxygen and biomass in the gasifier, they are classified according to the way air or oxygen is introduced in it. There are three types of gasifier as shown in Figure 9.2.

Figure 9.2 Types of fixed bed gasifiers
- Downdraft gasifiers: In the downdraft gasifier, the air is passed from the layers in the downdraft direction. Single throat gasifiers are mainly used for stationary applications, whereas double throat gasifier is used for varying loads as well as automotive purposes.
- Updraft gasifiers: Updraft gasifier has air passing through the biomass from bottom and the combustible gases come out from the top of the gasifier.
- Cross draft gasifiers: It is a very simple gasifier and is highly suitable for small outputs. With slight variation, almost all the gasifiers fall in the abovementioned categories.
The choice of one type of gasifier over other is dictated by the fuel, its final available form, its size, moisture content, and ash content. Table 9.2 lists the comparative features of various types of fixed bed gasifiers.
Table 9.2 Advantages and Disadvantages of Fixed Bed Gasifiers

- Fluidized bed gasifier: In fluidized bed gasifier, an inert material (such as sand, ash, or char) is utilized to make bed and that acts as a heat transfer medium
9.6 CHEMISTRY OF REACTION PROCESS IN GASIFICATION
Four distinct processes take place in a gasifier when fuel makes its way to gasification:
- Drying zone of fuel: In this zone, the moisture content of biomass is removed to obtain the dry biomass. Some organic acids also come out during the drying process. These acids give rise to corrosion of gasifiers.
- Pyrolysis zone: In this zone, the tar and other volatiles are driven off. The products depend upon temperature, pressure, residence time, and heat losses. However, following general remarks can be made about them.
- Up to the temperature of 200°C, only water is driven off.
- Between 200°C and 280°C carbon dioxide, acetic acid, and water are given off.
- The real pyrolysis, which takes place between 280°C and 500°C, produces large quantities of tar and gases containing carbon dioxide. Besides light tars, some methyl alcohol is also formed.
- Between 500°C and 700°C, the gas production is small and contains hydrogen.
- Combustion(oxidation) zone: In this zone, carbon from the fuel combust and forms carbon dioxide with the oxygen in the air by the reaction:
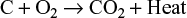
Because of the heat emitted during the reaction, the temperature rises until a balance between heat supply and heat loss occurs.
- Reduction zone: The hot gas passes through the reduction zone after the combustion zone. As there is no free oxygen in this zone that causes inflammable carbon dioxide gas to react with the carbon in the fuel and forms flammable carbon monoxide gas. This reaction is endothermic (demands heat) and occurs at temperature exceeding about 1,000°C. Carbon monoxide is the most important flammable elements in the produced gas obtained from the reduction reaction as
![]() (9.3)
(9.3)
Another important endothermic reaction in the reduction zone is the water–gas shift reaction. It is the reaction of water vapour and carbon to give carbon monoxide and hydrogen
![]() (9.4)
(9.4)
Both gasses are flammable, and the heating value of the gas is increased. If there is still surplus of water in the reduction zone, then carbon monoxide may react with water vapour and form carbon dioxide and hydrogen. This reaction is exothermic (emits heat) and decreases the heating value of the produced gas. The reaction is
![]() (9.5)
(9.5)
Equations (9.3) and (9.4) are main reduction reactions and being endothermic have the capability of reducing gas temperature. Consequently, the temperatures in the reduction zone are normally 800°C–1,000°C. The lower the reduction zone temperature (~700°C–800°C), lower is the calorific value of gas.
The gas also contains measurable amounts of particulate material and tar. The heating value of the gas ranges from 4,000 to 5,000 kJ/m3, which is a relatively low value when compared to the heating value of other gaseous fuels like natural gas.
The conversion efficiency of a gasifier is defined as the ratio of the heat content in the producer gas to the heat content in the biomass supplied and is usually around 75%.
Although there is a considerable overlap of the processes, each can be assumed to occupy a separate zone where fundamentally different chemical and thermal reactions take place.
Figure 9.3 shows schematically an updraft gasifier with different zones and their respective temperatures. Figure 9.4 for downdraft and Figure 9.5 for cross-draft also show these regions.
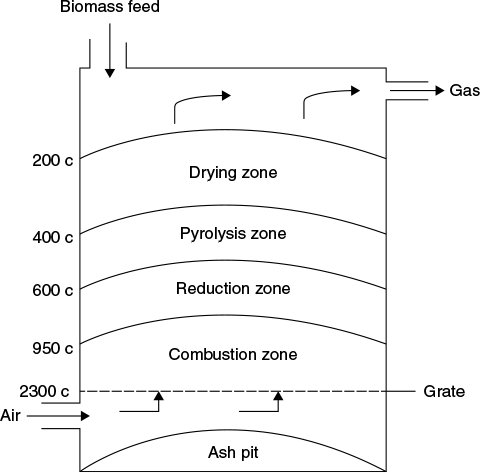
Figure 9.3 Updraft gasifier
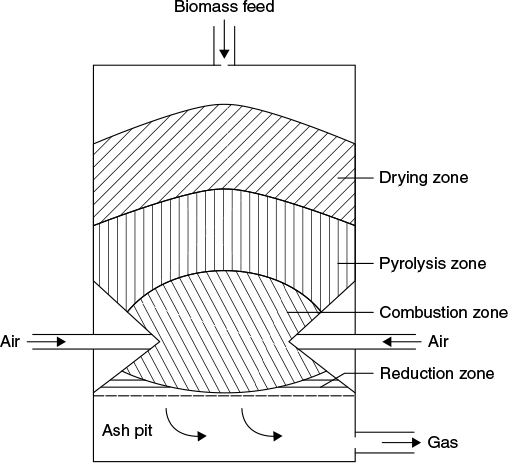
Figure 9.4 Downdraft gasifier
9.7 UPDRAFT GASIFIERS
The oldest and simplest type of gasifier is the counter current or updraft gasifier shown schematically in Figure 9.3. The air intake is at the bottom and gas leaves at the top (the counter current flow). The reactive agent is injected at the bottom of the reactor and ascends to the top, while the fuel is introduced at the top and descends to the bottom. The combustion reactions occur near the grate at the bottom that are followed by reduction reactions somewhat higher up in the gasifier. In the upper part of the gasifier, heating and pyrolysis of the feedstock occur as a result of heat transfer by forced convection and radiation from the lower zones. Gases, tar, and other volatile compounds are dispersed at the top of the reactor, while ash is removed at the bottom. The syngas typically contains high levels of tar, which must be removed or further converted to syngas for use in applications other than direct heating.
Updraft gasifiers are widely used to gasify biomass resources and generally use steam as the reactive agent, but slagging can be severe if high ash fuels are used. They are unsuitable for use with fluffy, low-density fuels.
These gasifiers are best suited for applications where moderate amounts of dust in the fuel gas are acceptable and a high flame temperature is required. Typical applications where the updraft gasifiers have been successfully used are as follows:
- Packaged boilers
- Thermal fluid heaters
- Aluminium melting/annealing furnaces
- All kinds of fryer roaster
9.8 DOWNDRAFT GASIFIER
In this gasifiers, the primary gasification air is introduced at or above the oxidation zone in the gasifier and the producer gas is removed at the bottom of the apparatus, so that fuel and gas move in the same direction, as schematically shown in Figure 9.4. The biomass feed (such as wood waste) and its gasification air both flow in the same downward direction through the gasifiers’ fuel bed.
The biomass feed is admitted at the top similar to the updraft gasifier. As the feed progresses down through the gasifier, it dries and its volatiles are pyrolysed. The char is directed into a reduced-diameter cylindrical throat section at the bottom of the gasifier. Gasification air is injected into the throat through openings in the throat wall. Due to the high temperatures existing at the throat section, tars and oils could be cracked, which tend to form in producer gas, particularly when the biomass is wetter than about 20% moisture content (wet basis). The producer gas leaves at the bottom of the gasifier. The start-up time of about 5–10 min is necessary to ignite and bring plant to working temperature with good gas quality is shorter than updraft gas producer.
Downdraft gasifiers are widely used in the following applications:
- Continuous baking ovens (bread, biscuits, and paint)
- Batch type baking oven (rotary oven for bread)
- Dryers and curing (tea, coffee, mosquito coil, and paper drying)
- Boilers
- Thermal fluid heaters
- Annealing furnaces
- Direct fired rotary kilns
- Internal combustion engines
9.9 CROSS-DRAFT GASIFIER
Figure 9.5 is a schematic representation of cross-draft gasifier. Unlike downdraft and updraft gasifiers, the ash bin, fire, and reduction zone in cross-draft gasifiers are separated.
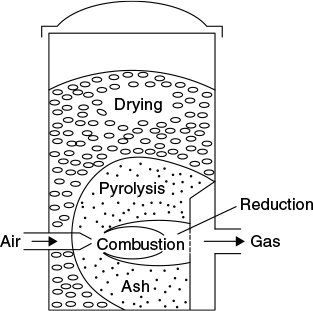
Figure 9.5 Cross-draft gasifier
These design characteristics limit the type of fuel for operation to low ash fuels such as wood, charcoal, and coke. The relatively high temperature in cross-draft gas producer has an obvious effect on gas composition such as high carbon monoxide, and low hydrogen and methane content when dry fuel like charcoal is used. Cross-draft gasifier operates well on dry air blast and dry fuel.
Typically, the gasifier is a vertical cylindrical vessel of varying cross section. The biomass is fed in at the top at regular intervals of time and is converted through a series of processes into producer gas and ash, as it moves down slowly through various zones of the gasifier.
9.10 FLUIDIZED BED GASIFICATION
Fluidized bed gasification has been successfully used to convert prepared wastes (i.e., wood wastes, bark, agricultural wastes, and RDF (Refused Derived Fuel) into a clean fuel gas that can be used to fire various types of industrial equipment. Past applications have included gasification of wastes to provide gas for dryers previously fired on natural gas.
The fluidized bed gasifier is illustrated schematically in Figure 9.6. This gasifier is an improved version of fixed bed gasifiers. The bed made of an inert material (such as sand, ash, or char) initially and it is heated and the fuel is introduced when the temperature has reached the appropriate level. The bed material transfers heat to the fuel and blows the reactive agent through a distributor plate at a controlled rate. Fluidized bed gasifiers have no distinct reaction zones (as in the case of fixed bed gasifiers) and drying, pyrolysis and gasification occur simultaneously. The fuel particles are introduced at the bottom of the reactor, very quickly mixed with the bed material and almost instantaneously heated up to the bed temperature. As a result of this treatment, the fuel is pyrolysed very fast, resulting in a component mix with a relatively large amount of gaseous materials. Further gasification and tar-conversion reactions occur in the gas phase.
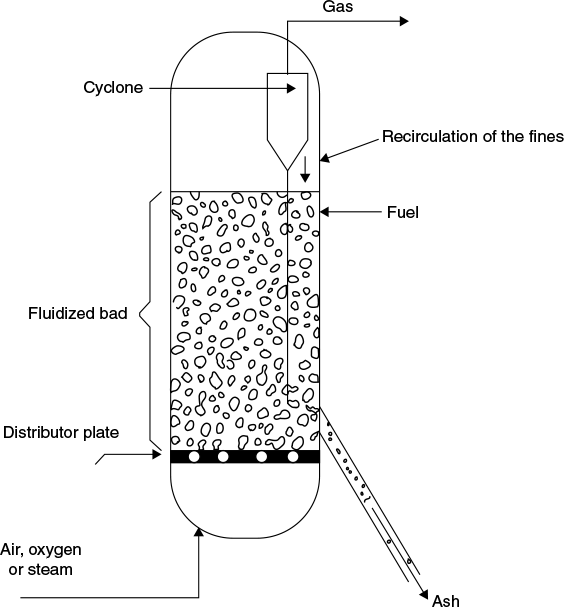
Figure 9.6 Fluidized bed gasifier
For biomass feeds which have high ash content and the ash has low melting point, fluidized bed combustion seems to gasify them.
9.10.1 Advantages and Benefits
The fluidized bed gasification process offers several substantial benefits when compared to simple burning processes and other forms of gasification.
9.10.1.1 Advantages
- Reduced cost of boiler or dryer or kiln operation by using wood and/or bark wastes rather than gas or oil.
- Reduced cost for additional steaming capacity when compared to new wood and or bark-fired boilers.
- Reduced dependency on external fuel sources for propane, natural gas, and oil.
9.10.1.2 Benefits
- High overall efficiency: High efficiency in the range of 70%–90% can be achieved. Moisture contents and the ash contents reduced the overall thermal efficiency of fluidized bed gasifier
- Fuel flexibility: The fluidized bed gasifiers have fuel flexibility and operate satisfactorily with highly variable feed materials. Ranging from coal, shredded wood and bark to sawdust fines, or lump wood with particle sizes of less than 4–6 cm. Thus, the various types of fuels generally available around lumber mills can be used in fluid bed gasifiers with good results.
- Highly reliable: The fluidized bed gasifier neither have moving grates nor other moving parts in the high temperature regions of the bed and hence they are highly reliable.
- Low purchase and installation costs: Air flow used in the gasifiers is comparatively low, and hence size of gasifier is small and compact. These permit systems to be completely shop fabricated and assembled on skids, thereby reducing purchase price and installed costs.
- Flexible operations: Fuel gas product of fluidized bed gasifier is easily applied to a variety of industrial processes including boilers, dry kilns, veneer dryers, or several pieces of equipment at once. Thus, they provide flexible operations.
- Low emissions: They are very low emission gasifiers and do not require exhaust clean up devices.
9.11 USE OF BIOMASS GASIFIER
The output of a biomass gasifier can be used for a variety of direct thermal applications such as cooking, drying, heating water, and generating steam. It can also be used as a fuel for internal combustion engines to obtain mechanical shaft power or electrical power.
If used as a fuel for internal combustion engines, it has to be cleaned first for complete removal of particulate material and tar. A cleaning system consisting of cyclone, a scrubber, and a filter is used for the purpose. If the engine is a spark-ignition engine, it can operate with producer gas alone. The gas is sucked from the gasifier and cleaner unit by the engine suction along with a proportionate amount of air. It is then compression-ignition engine, as it operates in the ‘dual-fuel’ mode. Here, the engine sucks in a mixture that is compressed and a small amount of diesel sprayed in. Combustion initiates with the diesel droplets and then spreads to the mixture of the gaseous fuel and air. The phrase ‘dual-fuel’ implies that both diesel and producer gas are simultaneously used.
India is one of the leading countries in the world in the field of biomass gasification. Biomass gasifier systems are available in a wide range of capacities and standard facilities for testing and evaluating gasifiers have been set up. For thermal applications, systems with outputs ranging from 60,000 to 5 × 106 kJ/h are available, while for electrical power generation, systems with outputs ranging from 3 to 500 kW are also available.
The largest biomass gasification system produces 5 × 106 kJ/h (1,450 kW) output in the thermal mode and 500 kW in the electric power generation mode. It uses biomass in the form of wood blocks (25 to 100 mm long and up to 70 mm in diameter) at the rate of 500 kg/h and produces 1,250 m3/h of gas. The internal combustion engine is a compression ignition engine operating in the dual-fuel mode. It uses only 25% of the diesel normally required by the engine if operating with diesel alone.
9.11.1 Liquid Fuels
When compared to gaseous fuels such as producer gas or biogas, liquid fuels are somewhat harder to obtain from biomass sources. One of the methods is the production of methanol from wood or straw. The process involves the gasification of plant matter followed by chemical synthesis. Another method is the conversion of certain food grains and crops such as sugarcane, maize, cassava and tapioca by fermentation into ethanol. When blended with petrol, ethanol is good alternate fuel for automotive engines. This fact has received considerable attention as a means of overcoming the oil crisis. However, if one examines the requirement of land for growing the agricultural products concerned, it is obvious that the method can be of substantial benefit only to a country having a large surplus of land. For this reason, Brazil has adopted this method on a large scale and produces significant amounts of ethanol for use as an alternate fuel. However, the position in India is quite different since the availability of land is limited.
In plants, algae and certain types of bacteria, the photosynthetic process results in the release of molecular oxygen and the removal of carbon dioxide from the atmosphere that is used to synthesize carbohydrates (oxygenic photosynthesis). Other types of bacteria use light energy to create organic compounds but do not produce oxygen (anoxygenic photosynthesis). Photosynthesis provides the energy and reduced carbon required for the survival of virtually all life on our planet, as well as the molecular oxygen necessary for the survival of oxygen consuming organism. In addition, the fossil fuels, currently being burned to provide energy for human activity, were produced by ancient photosynthetic organisms.
9.12 GASIFIER BIOMASS FEED CHARACTERISTICS
Most of gasifier manufacturers claim that a gasifier is available and can gasify any biomass feed. However, there is no such thing as a universal gasifier. A gasifier is in real sense very much biomass feed specific and it is tailored accordingly.
Following biomass feed characteristics or parameters dictate the quality and classification of gasifiers:
- Energy content of the fuel
- Bulk density
- Moisture content
- Dust content
- Tar content
- Ash and slogging characteristic
9.12.1 Energy Content and Bulk Density of Fuel
The higher the energy content and bulk density of fuel, the similar is the gasifier volume; as for one biomass fuel charge, power can be obtained for longer time duration.
9.12.2 Moisture Content
Moisture content is very trivial components of biomass fuels and it is determined by the type of fuel, its origin, and treatment. It is desirable to use fuel with low moisture content to minimize heat loss due to its evaporation.
Besides impairing the gasifier heat budget, high moisture content also puts load on cooling and filtering equipment by increasing the pressure drop across these units because of condensing liquid. Thus, in order to reduce the moisture content of fuel, some pre-treatment of fuel is required. Generally, desirable moisture content for fuel should be less than 20%.
9.12.3 Dust Content
All gasifier fuels produce undesirable dust that can clog the internal combustion engine and hence it has to be removed. The gasifier design should be such that it should not produce dust beyond specified limits.
The higher the dust produced, more is the load put on filters necessitating their frequent flushing and increased maintenance.
9.12.4 Tar Content
Tar is one of the most unpleasant constituents of the gas as it tends to deposit in the carburettor and intake valves causing sticking and troublesome operations. It is a product of highly irreversible process taking place in the pyrolysis zone. There are approximately 200 chemical constituents that have been identified in tar so far.
Very little research work has been done in the area of removing or burning tar in the gasifier so that relatively tar free gas comes out. Thus, the major effort has been devoted to cleaning this tar by filters and coolers.
9.12.5 Ash and Slagging Characteristics
The mineral content in the fuel that remains in oxidized form after complete combustion is usually called ash. The ash content of a fuel and the ash composition has a major impact on trouble-free operation of gasifier. Ash basically interferes with the gasification process in two ways:
- It fuses together to form slag and this clinker stops or inhibits the downward flow of biomass feed.
- Even if it does not fuse together, it shelters the points in fuel where ignition is initiated, and thus lowers the fuel’s reaction response.
Ash and tar removal are the two most important processes in gasification system for its smooth running. However, slagging can be overcome by two types of operation of gasifier:
- Low temperature operation that keeps the temperature well below the flow temperature of the ash.
- High temperature operation that keeps the temperature above the melting point of ash.
The first method is usually accomplished by steam or water injection, while the latter method requires provisions for tapping the molten slag out of the oxidation zone. Each method has its advantages and disadvantages and depends on specific fuel and gasifier design.
Keeping in mind the abovementioned characteristics of fuel, only two fuels have been thoroughly tested and proven to be reliable. They are charcoal and wood.
As charcoal is tar free and has relatively low ash content property, it was the preferred fuel during World War II and still remains so. However, there is a major disadvantage of charcoal in terms of energy. Charcoal is mostly produced from wood and in the conversion of wood to charcoal, about 50% of original energy is lost.
9.12.6 Biomass Feed (Fuel)
The major biomass sources presently used are as follows:
- Sugarcane and corn, wheat, sugar beet, sweet sorghum, and cassava to produce bioethanol.
- Rapeseed, sunflower seeds, soybean, canola, peanuts, jatropha, coconut, and palm oil for biodiesel production.
- Wide range of cellulosic materials (such as grassy crops, woody plants, by-products from the forestry and agricultural sector including wood residues, stems, and stalks and municipal wastes constitute the so-called second generation of feedstock).
- Wastes and residues constitute a large source of biomass. These include solid and liquid municipal wastes, manure, lumber and pulp mill wastes, and forest and agricultural residues.
Low water content (dried) biomass feedstock is burnt to generate heat and electricity. Wood wastes in the paper and pulp industries and bagasse from the sugarcane industry are used in ethanol fermentation.
A variety of raw materials that include agricultural wastes, municipal solid wastes, market garbage, and waste, water from food and fermentation industries (all organic materials containing carbohydrates, lipids, and proteins) are feedstock for anaerobic digester for methane production.
9.13 APPLICATIONS OF BIOMASS GASIFIERS
The main applications of biomass gasifier products are as follows:
- Motive power: Gasifier products are used to provide shaft power to industrial and agricultural equipment and machinery such as
- Diesel engine operation on dual or 100% modes.
- Water pumps
- Tractors, harvesters, etc.
- Running of high efficiency Stirling engines.
- Direct heat applications: Gasifiers heat has direct heat applications such as
- Drying of agricultural crop and food products such as large cardamom, ginger, rubber, and tea at low temperature range of about 85°C–125°C.
- Baking of tiles and potteries in the moderate temperature range of about 800°C–900°C.
- For melting metals and alloys in non-ferrous in the temperature range of 700°C–1,000°C.
- As boiler fuels provide steam or hot water for process industries such as silk reeling, dyeing, turmeric boiling, cooking, jiggery making, etc.
- Electrical power generation: Electric power generation from few kilowatts to hundreds of kilowatts either for local consumption or for grid power is being installed based on gasifier products. Small-scale electricity generation systems also provide an attractive alternative to electric supply company.
- Chemical production: Production of chemicals such as methanol and formic acid from producer gas.
9.14 COOLING AND CLEANING OF GAS
For efficient and effective use of gas for numerous applications, it should be cleaned of tar and dust, free from moisture content and cooled. Therefore, cooling and cleaning of the gas is one of the most important processes in the whole gasification system. The failure or the success of producer gas units depends completely on their ability to provide a clean and cool gas to the engines or for burners.
The temperature of gas coming out of generator is normally between 300°C and 500°C. The energy density of gas can be increased to a large extent by cooling it. Most coolers are gas to air heat exchangers where the cooling is done by free convection of air on the outside surface of heat exchanger. Some heat exchangers provide partial scrubbing of gas for the removal of moisture and tar contents. Thus, ideally, the gas going to an internal combustion engine should be cooled to nearly ambient temperature and shall be free from tar and moisture contents.
Normally, there are three types of filters used for cleaning of gas, as shown in Figure 9.7, which is schematically a downdraft gasification system with cleaning and cooling train.
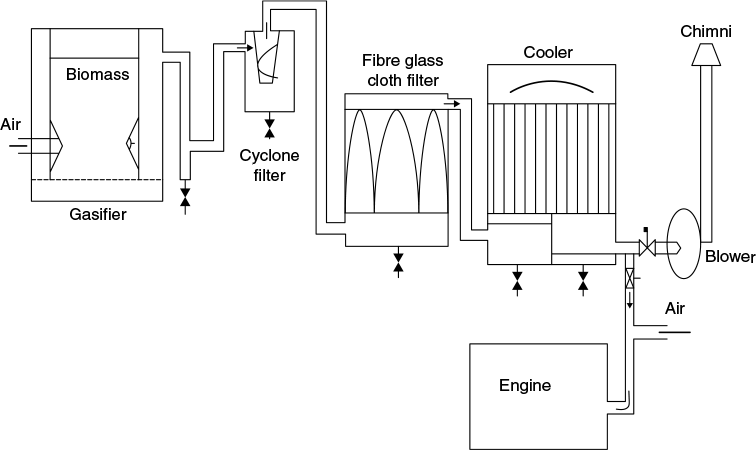
Figure 9.7 Schematic diagram of producer gas plant
They are classified as dry, moist, and wet.
- Cyclone filters: They are designed according to the rate of gas production and its dust content. They are useful for particle size of 5 μm and greater. Since 60%–65% of the producer gas contains particles above 60 μm in size, the cyclone filter is an excellent cleaning device.
- Wet scrubber: Even after cyclone filtering, the gas still contains fine dust, particles, and tar. It is further cleaned by passing through a wet scrubber where gas is washed by water in countercurrent mode. The scrubber also acts like a cooler, from where the gas goes to cloth or cork filter for final cleaning.
- Cloth filters: It is a fine filter. Any condensation of water on it stops the gas flow because of an increase in pressure drop across it. Thus, in quite a number of gasification systems, the hot gases are passed through the cloth filter, and then only do they go to the cooler. Since the gases are still above the dew point, no condensation takes place in filter.
SUMMARY
- Biomass gasification offers the most attractive alternative energy system for agricultural purpose
- Most preferred fuels for gasification have been charcoal and wood. However biomass residues are the most appropriate fuels for on-farm systems and offer the greatest challenge to researchers and gasification system manufacturers.
- Very limited experience has been gained in gasification of biomass residues.
- Most extensively used and researched systems have been based on downdraft gasification. However it appears that for fuels with high ash content fluidized bed combustion may offer a solution. At present no reliable and economically feasible systems exist.
- Biggest challenge in gasification systems lies in developing reliable and economically cheap cooling and cleaning trains.
- Maximum usage of producer gas has been in driving internal combustion engine, both for agricultural as well as for automotive uses. However, direct heat applications like grain drying is very attractive for agricultural systems.
- A spark ignition engine running on producer gas on an average produces 0.55–0.75 kWh of energy from 1 kg of biomass.
- Compression ignition (diesel) engines cannot run completely on producer gas. Thus to produce 1 kWh of energy they consume 1 kg of biomass and 0.07 l of diesel. Consequently, they effect 80%–85% diesel saving.
- Future applications like methanol production using producer gas in fuel cell are under investigation.
REVIEW QUESTIONS
- What is the meaning of biomass? Further, discuss its multipurpose utilization
- How biomass conversion takes place?
- State the names of raw biomass from waste and cultivated crops.
- State the significant aquatic biomass resources.
- State the important forest biomass resources
- State the important rural biomass.
- State the names of various agricultural wastes used in Fluidized Bed Combustion Boiler.
- Discuss the applications of aquatic energy sources.
- What are the basic steps involved in Waste Recycling?
- Describe the entire recycling route from the collection of waste to final supply of recycle material.
- Write the applications of biomass gasifiers.
- Explain issues in biomass energy productions.
- Classify and explain methods of obtaining energy from biomass.
- Define biomass gasification. Further, explain the average energy conversion efficiency of gasifiers.
- What is producer gas? Further, discuss its properties.
- List different types of gasifiers and compare them.
- State and explain processes of biomass gasification. Further, define average energy conversion efficiency of gasifiers.
- Draw the schematic representation of either of the following gasifiers. Further, explain their working and application areas.
- Updraft gasifier
- Downdraft gasifier
- Cross-draft gasifier
- Fluidized bed gasifier
- State and explain parameters that dictate the quality and classification of gasifiers.
- Describe the advantages and benefits of fluidized bed gasifier?
