1
Introduction, Vision, and Opportunities
Maria E. Holuszko1, Denise C. R. Espinosa2, Tatiana Scarazzato3, and Amit Kumar1
1NBK Institute of Mining Engineering, University of British Columbia, 6350, Stores Road, Vancouver, BC V6T 1Z4, Canada
2University of São Paulo, Polytechnic School, Department of Chemical Engineering, Av. Prof. Luciano Gualberto, 380 – Butantã, São Paulo – SP 05508-010, Brazil
3Federal University of Rio Grande do Sul, Department of Materials, 9500, Av. Bento Gonçalves, Porto Alegre – RS, 91509-900, Brazil
1.1 Background
The concept of sustainability defined by The United Nations Organization in 1987, which is valid even today, is based on the idea of “meeting the needs of the present without compromising the ability of future generations to meet their own needs” (Nations 2019). Such a concept was complemented in The Johannesburg Declaration on Sustainable Development in which the three pillars of sustainability were defined: economic, environmental, and social development (Comission 2002).
Notwithstanding, the world currently faces a transition between the third and the fourth industrial revolutions, which began about five decades ago and has transformed our way of living. Also known as the Information Revolution, this period has been marked by swift advances in computer technologies, massive popularization of high-technology devices, and the growth of artificial intelligence (Carvalho et al. 2018; Rai and Lal 2000). The technological revolution brought up the creation of lithium-ion batteries, touchscreen devices, supercomputers, photovoltaic panels, and nanocomposites, and practically revolutionized the way the society interacts, the way energy is stored, and the advanced materials field for all industrial sectors.
Electrical and electronic equipment is one of the major consumers of metals such as copper, gold, silver, and iron. Namias (2013) suggested that electronic devices can contain up to 60 different elements that could be valuable or hazardous. Natural Resources Canada (2019) showed that globally 18% of aluminum, 31% of copper, 9.5% of gold, 9% of platinum group metals, and 24% of rare-earth elements were used in electrical and electronic equipment manufacturing in 2017. In the United States of America, 9% of total aluminum, 21% of beryllium, 19% of copper, 40% of gold, and 26% of silver were used in the electrical and electronic equipment manufacturing industry in 2019 (U.S. Geological Survey 2019). BullionStreet (2012) showed that approximately 290 tonnes of gold and 6800 tonnes of silver are consumed by the electronic industry every year. In the current scenario, the new manufacturing industry became dependent on less-known raw materials and increased the extraction of common metals from ores simultaneously. Indium, for example, despite being discovered in 1863, was found to be industrially applicable only in 1934. The use of indium in thin-film coatings, mainly as indium-tin-oxide compound (ITO) in liquid crystal display screens, increased its world consumption over 1000% since 1993 (Alfantazi and Moskalyk 2003).
Rare-earth elements (REEs) are also widely used in digital technologies such as disc drivers and communication systems but also in batteries and fuel cells for hydrogen storage, catalysts, light-emitting diodes (LEDs), and fluorescent lighting. Back in 1950, the applications of REE in magnets of electric and electronic equipment were already known. Nevertheless, until 2010 their recycling rate was lower than 1% due to their relatively low prices (Gunn 2013). Between 2010 and 2015, the demand for REE surpassed its supply and continuously increased. As the production is almost totally held by few countries, the recycling of REE has become a paramount concern (Edahbi et al. 2019).
1.2 E-Waste
With the development of new technologies, especially in laptops, cellphones, and tablets, older technologies are getting obsolete, reducing the lifespan of electrical and electronics products and thus contributing to a higher rate of waste generation. As a result, close to 1 billion devices will be discarded within 4–5 years. The discarded electric and electronic equipment or their parts are considered e-waste. The European Commission Directive 2008/98/EC (2008) and the European Union Directive 2012/19/EU (2012) described e-waste as:
any electrical or electronic equipment which is waste (substance or object which the holder discards or intends or is required to discard), including all components, sub-assemblies, and consumables which are part of the product at the time of discarding.
Based on the definition of e-waste, the electrical or electronic equipment (EEE) itself was divided into six (6) classes in the Directive 2012/19/EU (The European Union 2012). These categories with the items (not limited to) in the categories are listed as,
- Temperature-exchange equipment: refrigerators, freezers, air conditioning equipment and, heat pumps
- Screens, monitors, and equipment containing screens (surface > 100 cm2): screens, televisions, LCD photo frames, monitors, laptops, and notebooks
- Lamps: fluorescent lamps, high-intensity discharge lamps, including high-pressure sodium lamps and metal halide lamps, low-pressure sodium lamps and LED lamps
- Large equipment (external dimensions > 50 cm): washing machines, dryers, dishwashers, electric stoves, musical equipment, large computer mainframes, large printing machines, copying equipment, large coin-slot machines, large medical devices, large automatic dispensers, and photovoltaic panels
- Small equipment (external dimensions < 50 cm): vacuum cleaners, appliances for sewing, luminaires, microwaves, irons, toasters, electric kettles, clocks and watches, electric shavers, scales, radio, video cameras, electrical and electronic toys, sports equipment, smoke detectors, heating regulators, thermostats, small electrical and electronic tools, small medical devices, and small automatic dispensers
- Small IT and telecommunication equipment (external dimension < 50 cm): mobile phones, GPS, pocket calculators, routers, personal computers, printers, and telephones
This electronic waste (discarded electronics) has been a growing concern around the world. The total e-waste generated around the globe in 2019 was 53.6 million tonnes and is expected to reach 74 million tonnes in 2030. The waste generated per capita increased from 6.1 kg per inhabitant in 2016 to 7.3 kg per inhabitant in 2019 (Forti et al. 2020). Wahlen (2019) reported that under the business-as-usual case, the total e-waste generation would increase to 120 million tonnes by 2050. The growth rate of e-waste generation has been reported to be 3–5% by Cucchiella et al. (2015), 3–4% by Baldé et al. (2017) and Aaron (2019), and as high as 8% by LeBlanc (2018). According to Transparency Market Research report (2017), the global e-waste market is anticipated to increase at a compound annual growth rate of 5.6% by volume from 2016 to 2026.
The fate of the e-waste can be described by the simplified diagram shown in Figure 1.1. The primary focus of any country or organization should be the collection and recycling of e-waste. However, not all the e-waste is collected, and a portion of the e-waste stream is disposed of in landfills. The collected materials are sent for recycling, and the high-value components such as metals and high-value plastics are fed back to the manufacturing stream, whereas low-value materials are disposed of in landfills.
The primary focus of any country or organization should be the collection and recycling of e-waste. However, not all the e-waste is collected, and a portion of the e-waste stream is disposed of in landfills. The collected materials are sent for recycling, and the high-value components such as metals and high-value plastics are fed back to the manufacturing stream, whereas low-value materials are disposed of in landfills. The e-waste collection volume must be increased to boost the circular economy in any part of the world, and the waste stream after the recycling process has to be studied simultaneously for its potential recovery and usage so that the fractions to be disposed of are minimized.
E-waste recycling decreases the amount of extracted raw materials from ores and solid waste inadequate disposal. The recycling routes must also be technically and economically feasible. Given the added value of precious metals and critical metals found in the majority of e-waste, such requirements are not difficult to be fulfilled. Baldé et al. (2017) estimated the amount of various elements and materials present in e-waste. It showed that the total contained/potential value of selected metal and materials present in e-waste was US$ 57 billion in 2019 (Forti et al. 2020). Figure 1.2 shows a breakdown of the various metals and materials present in e-waste with their total amount and estimated values. It should be noted that the estimated value depicted in Figure 1.2 represents an ideal-case scenario of 100% collection and metal recovery and without accounting for costs associated with collection and recycling. It indicates the economic opportunity for e-waste recycling.

Figure 1.1 Simplified flow of EEE products.

Figure 1.2 Estimated value of materials present in e-waste.
It should also be taken into consideration that the concentration of metals in the e-waste is significantly higher than that of a conventional mining operation. The global ore grade has been decreasing, and the increased global metal demand has forced mining operations to increase the plant throughput and excavate more complex and fine-grained ore deposits (Lèbre and Corder 2015).
Table 1.1 shows the concentration of various metals in different types of e-waste and an average ore body. Calvo et al. (2016) summarized that the global average copper grade for run-of-mine ore is ∼0.62% and will decline in the coming years due to the exhaustion of high-grade mines. AME Research (2018) showed that the average copper grade has decreased from 0.74% in 2005 to 0.59% in 2017, with a compound annual decline rate of 1.8%. The global average gold grade of all the deposits was 1.01 g/t in 2013 (Desjardins 2014). The highest gold grade for the underground operation was 21.5 ppm in Fosterville, Australia, whereas for open pit was 7.60 ppm in Way Linggo, Indonesia (Basov 2018). The average output of top-six silver mines was 7.6 oz (215 g) per tonne in 2012 and has dropped to 4–5 oz per ton in 2017 (McLeod 2014; Money Metals Exchange 2018). The palladium grade reported in Table 1.1 is based on the average palladium grade of the Lac des Iles Mine Property in Northern Ontario. It indicates that the average metal grade present in e-waste is significantly higher than conventional mines and thus provides the opportunity for the extraction/urban mining.
Table 1.1 Average metal content in various waste EEE and typical ore.
| Copper | Aluminum | Iron | Gold | Silver | Palladium | |
|---|---|---|---|---|---|---|
| % | % | % | ppm | ppm | ppm | |
| Air conditioner | 6–19 | 7–9 | 46 | 15 | 58 | – |
| Desktop | 7–20 | 2–4 | 18–47 | 46–240 | 207–570 | 18–25 |
| Laptop | 1–19 | 1–2 | 20 | 32–630 | 190–1100 | 19 |
| Mobile phone | 10–33 | 3 | 5 | 30–1500 | 2000–3800 | 300–1700 |
| Printed circuit board | 12–19 | 2–8 | 0–11 | 29–1120 | 100–5200 | 33–220 |
| Refrigerator | 3–17 | 1–2 | 48–50 | 44 | 42 | – |
| Television | 1–21 | 1–15 | 13–43 | 5–300 | 120–600 | 0–20 |
| Washing machine | 3–7 | 0–3 | 52–53 | 17 | 51 | – |
| e-Waste (average) | 12–35 | 1.5–5 | 5–11 | 30–350 | 80–1000 | 30–200 |
| Typical ore | 0.5–3 | 20–24 | 30–60 | 0.5–10 | 5–10 | 1–10 |
Sources: Based on Bizzo et al. (2014), Calvo et al. (2016), Desjardins (2014), Işıldar (2016), Bizzo et al. (2014), Calvo et al. (2016), Desjardins (2014), Fornalczyk et al. (2013), Işıldar (2016), Khaliq et al. (2014), Liu (2014), McLeod (2014), Namias (2013), North American Palladium Ltd. (2018), Shah et al. (2014), Tickner et al. (2016), Zeng et al. (2016).

Figure 1.3 Average percent weight of some common metals found in PCB, photovoltaic modules, and HD magnets (Caldas et al. 2015; Dias et al. 2016, 2018; Kasper et al. 2011; München et al. 2018; München and Veit 2017; Petter et al. 2014; Rozas et al. 2017; Sant’ana et al. 2013; Silvas et al. 2015; Stuhlpfarrer et al. 2015; Yamane et al. 2011).
Sources: Based on Caldas et al. (2015), Dias et al. (2016), and Kasper et al. (2011).
As depicted in Figure 1.3, photovoltaic modules present a high percent weight of a single element (aluminum), while printed circuit boards are composed of a mixture of different metals, mainly copper, iron, aluminum, tin, zinc, and nickel. It is estimated that printed circuit boards (PCBs) may contain an average of 18 elements from the periodic table (Caldas et al. 2015; Kasper et al. 2011; Petter et al. 2014; Rozas et al. 2017; Sant’ana et al. 2013; Silvas et al. 2015; Yamane et al. 2011). Remarkably, hard disk magnets, although they may contain high amounts of iron, also present exciting amounts of rare-earth elements, mainly neodymium, praseodymium, and dysprosium (München and Veit 2017).
Metals that are present in smaller amounts may also be economically advantageous to be recovered. Gold and silver in cell phones, for example, represent about 0.06% and 0.045% in weight, respectively (Caldas et al. 2015; Sant’ana et al. 2013). Considering the number of cell phones worldwide (Kreyenhagen 2018), it can be estimated that almost 300-ton gold is present only in cellphone devices.
The added value of such metals supports their recycling even if they are present in low percent weight. Photovoltaic modules, for example, although present less than 1% of silver in their composition (Figure 1.3), may be economically feasible to be recycled, as shown in Figure 1.4.
From Figure 1.4, it becomes evident that the establishment of feasible recycling routes may be advantageous for reclaiming metals in either high or low amounts. This is undoubtedly challenging, as e-waste from distinct origins would present different compositions and structures. In a microscopic vision, metals and other elements may be bonded with each other in many possibilities, which may require different techniques to achieve their extraction and recovery. Thus, recycling routes must be versatile. Electric and electronic equipment are constantly being improved, and their chemical composition may change both the required process and the obtained materials. Lithium-ion batteries (LIBs) are a good example of continuous improvement. Cathodes from LIBs are typically composed of lithium, cobalt, manganese, and nickel oxides (Blomgren 2017; Zhao et al. 2019). Because cobalt is considered a critical metal, novel cathode materials are being developed, such as sulfur-based cathodes, which eliminate the need for cobalt (Li et al. 2018). High-efficiency anodes are also being developed to increase the performance of LIBs, such as titanium-niobium based anodes for automotive applications (Takami et al. 2018). Considering the speed of technological evolution, in a few decades from now, the composition of obsolete lithium-ion batteries may present a substantial change.
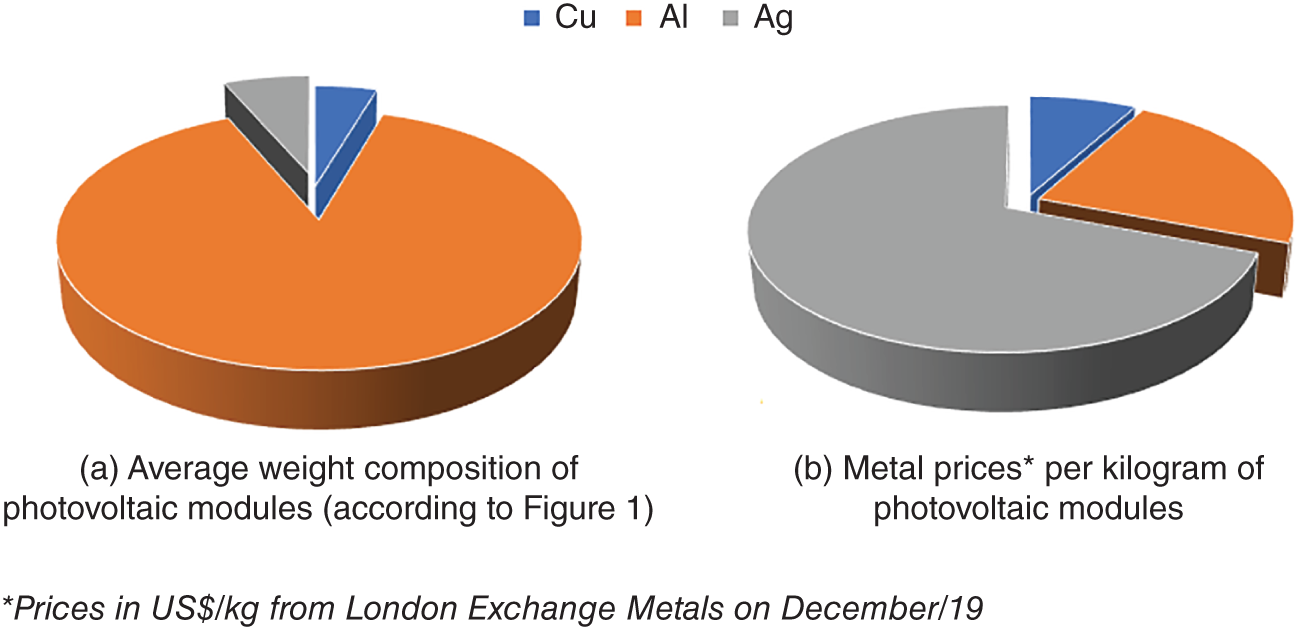
Figure 1.4 (a) Composition of photovoltaic modules illustrated as kilograms of each metal per kilogram of photovoltaic module. (b) Value of each metal per kilogram of photovoltaic modules.
Recycling processing routes are typically hydrometallurgical, pyrometallurgical, or hydro-pyrometallurgical. Each of them presents both advantages and disadvantages, and all of them may produce toxic tailings, which must be considered. The pyrometallurgical recycling routes consist of several processes that, among others, include smelting, combustion, pyrolysis, molten salt processing, and pyro-chemical techniques. As for drawbacks to the pyrolysis process, there is usually the release of toxic gases and halogen formation arising from fire retardants and plastic mixtures that compose the waste electrical or electronic equipment (WEEE) scrap. For the efficient recovery of metals, some modern methods, such as vacuum pyrolysis, the molten salt process, and pyro-chemistry, appear to be promising candidates because of their innovative solutions for environmental issues and recovery efficiency. Nevertheless, they still need more scientific and technical contributions coupled with industrial validation (Zhang et al. 2015).
Hydrometallurgical processing involves a sequence of methods for producing metals and metal compounds from aqueous media. In general, the process is composed of leaching techniques, followed by purification and recovery of metals, resulting in the desired product. Such a product may be either metals or alloys and compounds containing the metals of interest, such as oxides (Gupta 2003). Precipitation, hydrolysis, electrochemistry, conversion, complexation, solvation, and ionic dissociation are often used in different processing steps. However, such routes often require the use of aggressive and concentrated reactants for leaching steps. The treatment of the generated waste belongs to the development of a hydrometallurgical route. In addition, some processes still present low efficiency. To solve such issues, the scientific community is currently focusing on the development of less-toxic processing routes, using, for example, ionic liquids, supercritical fluids, and organic acids, aiming at achieving optimum extraction rate using greener chemicals.
Nevertheless, another important matter is the efficient collection of the disposed of WEEE to get to recycling industries. The incitement of reversal logistics and circular economy concepts must be improved and optimized to achieve satisfactory rates of recycled WEEE. In this sense, the establishment of strict legislation and efficient management are as well important. In developing countries, for example, WEEE recycling is still incipient, there is little legislation, and incentives are low. Even in developed countries, there is still much to be improved.
These legislations and programs are essential to increase awareness and boost the collection rate. The e-waste management programs also provide collection targets, summarize, and report the collection volumes, which helps to understand the performance and provide better planning tools for the future. An efficient e-waste management program could increase the collection rates, provide a better estimation of e-waste collection, and would also provide a better understanding of the steps required to improve e-waste collection and recycling and thus promote circular economy. At the same time, a higher collection rate in tandem with an efficient recycling system that not only recovers metals but also provides a solution for the nonmetals would be necessary to increase the e-waste circularity.
Therefore, it becomes evident that e-waste recycling is an interdisciplinary and multidisciplinary theme, as depicted in Figure 1.5. Technical, economic, legislative, social, and environmental aspects are involved throughout the life cycle of all-electric and electronic equipment, including recycling after their disposal. This book seeks to provide an overview of all aspects of a sustainable future.
1.3 Outline
To fully understand the e-waste, this book presents various sections to describe different aspects of e-waste, e-waste management systems and involved technologies, and other related disciplines.
Chapter 2 presents an overview of e-waste management practices adopted in developed and developing countries. The differences in the availability of regulations in a country directly impact the fate of e-waste in that country.
In Chapter 3, the regulations related to the transboundary movement of e-waste around the world are discussed. The import and export of e-waste is another major challenge in the e-waste management system.

Figure 1.5 Multidisciplinary aspects of e-waste recycling.
Various models and methods used for the quantification of total e-waste globally are presented in Chapter 4. The success of an e-waste recycling or disposal facility depends on the annual throughput to plant.
Chapter 5 emphasizes the materials used in the manufacturing of electronic devices. The determination of type and quantity of the materials used in the manufacturing process define the technologies adapted for recycling.
A detailed view of recycling technologies used in e-waste processing is presented in Chapters 6–10.
Chapter 11 presents insights into the life cycle analysis of obsolete electric and electronic equipment. The life cycle analysis is an essential tool for quantifying the environmental impact of e-waste.
Finally, the future of electronic devices and e-waste is discussed in Chapters 12 and 13, focusing on innovative aspects of manufacturing electronic devices, green chemistry, and circular economy.
References
- Aaron, J. (2019). Why is E-waste management important in 2019? https://get-green-now.com/e-waste-management (accessed 15 March 2021).
- Alfantazi, A.M. and Moskalyk, R.R. (2003). Processing of indium: a review. Minerals Engineering 16 (8): 687–694. https://doi.org/10.1016/S0892-6875(03)00168-7.
- AME Research (2018). Copper – declining copper ore grades. https://aus.amegroup.com/Website/FeatureArticleDetail.aspx?faId=437 (accessed 15 March 2021).
- Baldé, C. P., Forti, V., Gray, V., et al. (2017). The global E-waste monitor – 2017. https://www.itu.int/en/ITU-D/Climate-Change/Documents/GEM2017/Global-E-waste Monitor 2017.pdf (accessed 15 March 2021).
- Basov, V. (2018). Highest-grade gold mines in 2017. https://www.mining.com/highest-grade-gold-mines-2017 (accessed 15 March 2021).
- Bizzo, W., Figueiredo, R., and de Andrade, V. (2014). Characterization of printed circuit boards for metal and energy recovery after milling and mechanical separation. Materials 7 (6): 4555–4566. https://doi.org/10.3390/ma7064555.
- Blomgren, G.E. (2017). The development and future of lithium ion batteries. Journal of the Electrochemical Society 164 (1): A5019–A5025. https://doi.org/10.1149/2.0251701jes.
- BullionStreet (2012). Electronics industry uses 320 tons of gold, 7500 tons of silver annually. http://www.bullionstreet.com/news/electronics-industry-uses-320-tons-of-gold7500-tons-of-silver-annually/2255 (accessed 15 March 2021).
- Caldas, M.P.K., Moraes, V.T., De Junca, E. et al. (2015). Recuperação De Prata : Estudo De Uma Rota Hidrometalúrgica recycling of printed circuit boards aiming silver recovery. Tecnologia Em Metalurgia, Materiais e Mineração 12: 102–108.
- Calvo, G., Mudd, G., Valero, A., and Valero, A. (2016). Decreasing ore grades in global metallic mining: a theoretical issue or a global reality? Resources 5 (4): 36. https://doi.org/10.3390/resources5040036.
- Carvalho, N., Chaim, O., Cazarini, E., and Gerolamo, M. (2018). Manufacturing in the fourth industrial revolution: a positive prospect in sustainable manufacturing. Procedia Manufacturing 21: 671–678. https://doi.org/10.1016/j.promfg.2018.02.170.
- Cucchiella, F., D’Adamo, I., Lenny Koh, S.C., and Rosa, P. (2015). Recycling of WEEEs: an economic assessment of present and future e-waste streams. Renewable and Sustainable Energy Reviews 51: 263–272. https://doi.org/10.1016/j.rser.2015.06.010.
- Desjardins, J. (2014). Global gold mine and deposit rankings 2013. http://www.visualcapitalist.com/global-gold-mine-and-deposit-rankings-2013 (accessed 15 March 2021).
- Dias, P., Javimczik, S., Benevit, M. et al. (2016). Recycling WEEE: extraction and concentration of silver from waste crystalline silicon photovoltaic modules. Waste Management 57: 220–225.
- Dias, P., Schmidt, L., Gomes, L.B. et al. (2018). Recycling waste crystalline silicon photovoltaic modules by electrostatic separation. Journal of Sustainable Metallurgy 4 (2): 176–186. https://doi.org/10.1007/s40831-018-0173-5.
- Edahbi, M., Plante, B., and Benzaazoua, M. (2019). Environmental challenges and identification of the knowledge gaps associated with REE mine wastes management. Journal of Cleaner Production 212: 1232–1241. https://doi.org/10.1016/j.jclepro.2018.11.228.
- Fornalczyk, A., Willner, J., Francuz, K., and Cebulski, J. (2013). E-waste as a source of valuable metals. Archives of Materials Science and Engineering 63 (2): 87–92. https://www.researchgate.net/publication/260706030_E-waste_as_a_source:of_valuable_metals (accessed 15 March 2021).
- Forti, V., Baldé, C. P., Kuehr, R., and Bel, G. (2020). Global E-waste Monitor 2020: Quantities, flows and the circular economy potential. https://www.itu.int/en/ITU-D/Environment/Documents/Toolbox/GEM_2020_def.pdf (accessed 15 March 2021).
- Gunn, G. (2013). Critical Metals Handbook, 1–439. Wiley https://doi.org/10.1002/9781118755341.
- Gupta, C.K. (2003). Chemical Metallurgy Principles and Practice. Wiley.
- Işıldar, A. (2016). Biological Versus Chemical Leaching of Electronic Waste for Copper and Gold Recovery. Université Paris-Est Marne-la-Vallée https://tel.archives-ouvertes.fr/tel-01738056/document (accessed 15 March 2021).
- Kasper, A.C., Berselli, G.B.T., Freitas, B.D. et al. (2011). Printed wiring boards for mobile phones: characterization and recycling of copper. Waste Management 31 (12): 2536–2545. https://doi.org/10.1016/j.wasman.2011.08.013.
- Khaliq, A., Rhamdhani, M., Brooks, G., and Masood, S. (2014). Metal extraction processes for electronic waste and existing industrial routes: a review and Australian perspective. Resources 3 (1): 152–179. https://doi.org/10.3390/resources3010152.
- Kreyenhagen, J. (2018). Why Catering to Mobile Users is Vita for Today’s Services. Forbes Communications Council.
- LeBlanc, R. (2018). E-waste recycling facts and figures. https://www.thebalancesmb.com/e-waste-recycling-facts-and-figures-2878189 (accessed 15 March 2021).
- Lèbre, É. and Corder, G. (2015). Integrating industrial ecology thinking into the management of mining waste. Resources 4 (4): 765–786. https://doi.org/10.3390/resources4040765.
- Li, G., Chen, Z., and Lu, J. (2018). Lithium-sulfur batteries for commercial applications. Chem 4 (1): 3–7. https://doi.org/10.1016/j.chempr.2017.12.012.
- Liu, S. (2014). Analysis of electronic waste recycling in the United States and potential application in China [Columbia University]. https://secureservercdn.net/198.71.233.185/epm.300.myftpupload.com/wp-content/uploads/2020/10/FINAL_Shumeng_Thesis_Dec-20_2014.pdf.
- McLeod, C. (2014). Top primary silver miners facing declining ore grades. http://investingnews.com/daily/resource-investing/precious-metals-investing/silver-investing/top-primary-silver-miners-facing-declining-ore-grades (accessed 15 March 2021).
- Money Metals Exchange. (2018). World's largest silver mines: suffer falling ore grades & rising costs/seeking alpha. https://seekingalpha.com/article/4141630-worlds-largest-silver-mines-suffer-falling-ore-grades-and-rising-costs (accessed 15 March 2021).
- München, D.D., Bernardes, A.M., and Veit, H.M. (2018). Evaluation of neodymium and praseodymium leaching efficiency from post-consumer NdFeB magnets. Journal of Sustainable Metallurgy 4 (2): 288–294. https://doi.org/10.1007/s40831-018-0180-6.
- München, D.D. and Veit, H.M. (2017). Neodymium as the main feature of permanent magnets from hard disk drives (HDDs). Waste Management 61: 372–376. https://doi.org/10.1016/j.wasman.2017.01.032.
- Namias, J. (2013). The Future of Electronic Waste Recycling in the United States: Obstacles and Domestic Solutions. New York, United States: Columbia University.
- Natural Resources Canada (2019). Minerals and metals facts. https://www.nrcan.gc.ca/minerals-metals-facts/20507 (accessed 15 March 2021).
- North American Palladium Ltd. (2018). North American Palladium completes a new feasibility study featuring a major expansion of underground reserves. https://www.nap.com/investors/news-releases/news-releases-details/2018/North-American-Palladium-Completes-a-New-Feasibility-Study-Featuring-a-Major-Expansion-of-Underground-Reserves/default.aspx (accessed 15 March 2021).
- Petter, P.M.H., Veit, H.M., and Bernardes, A.M. (2014). Evaluation of gold and silver leaching from printed circuit board of cellphones. Waste Management 34 (2): 475–482. https://doi.org/10.1016/j.wasman.2013.10.032.
- Rai, L.P. and Lal, K. (2000). Indicators of the information revolution. Technology in Society 22 (2): 221–235. https://doi.org/10.1016/S0160-791X(00)00006-3.
- Rozas, E.E., Mendes, M.A., Nascimento, C.A.O. et al. (2017). Bioleaching of electronic waste using bacteria isolated from the marine sponge Hymeniacidon heliophila (Porifera). Journal of Hazardous Materials 329: 120–130. https://doi.org/10.1016/j.jhazmat.2017.01.037.
- Sant’ana, H.B.S., Moura, F.J., and Veit, H.M. (2013). Caracterização Físico-Química De Placas De Circuito Impresso De Aparelhos De Telefone Celular. Tecnologia Em Metalurgia Materiais e Mineração 10 (3): 231–238. https://doi.org/10.4322/tmm.2013.033.
- Shah, M.B., Tipre, D.R., and Dave, S.R. (2014). Chemical and biological processes for multi-metal extraction from waste printed circuit boards of computers and mobile phones. Waste Management & Research 32 (11): 1134–1141. https://doi.org/10.1177/0734242X14550021.
- Silvas, F.P.C., Jiménez Correa, M.M., Caldas, M.P.K. et al. (2015). Printed circuit board recycling: physical processing and copper extraction by selective leaching. Waste Management 46: 503–510. https://doi.org/10.1016/J.WASMAN.2015.08.030.
- Stuhlpfarrer, P., Luidold, S., and Antrekowitsch, H. (2015). Recycling of Nd2Fe14B magnets. World of Metallurgy - ERZMETALL 68 (5): 278–285.
- Takami, N., Ise, K., Harada, Y. et al. (2018). High-energy, fast-charging, long-life lithium-ion batteries using TiNb2O7 anodes for automotive applications. Journal of Power Sources 396 (June): 429–436. https://doi.org/10.1016/j.jpowsour.2018.06.059.
- The European Commission (2008). Directive 2008/98/EC of the European Parliament and of the council of 19 November 2008 on waste and repealing certain directives. Official Journal of the European Union L 312: 3–30. https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32008L0098&from=EN (accessed 15 March 2021).
- The European Union (2012). Directive 2012/19/EU of the European Parliament and of the Council of the 4 July 2012 on waste electrical and electronic equipment (WEEE). Official Journal of the European Union L (June) https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32012L0019&from=EN (accessed 15 March 2021).
- Tickner, J., Rajarao, R., Lovric, B. et al. (2016). Measurement of gold and other metals in electronic and automotive waste using gamma activation analysis. Journal of Sustainable Metallurgy 2 (4): 296–303. https://doi.org/10.1007/s40831-016-0051-y.
- Transparency Market Research. (2017). E-scrap and printed circuit board (PCB) E-scrap market. https://www.transparencymarketresearch.com/e-scrap-printed-circuit-board-e-scrap-market.html (accessed 15 March 2021).
- U.S. Geological Survey (2019). Mineral Commodity Summaries. U.S. Geological Survey https://doi.org/10.3133/70202434.
- UN Department of Economic and Social Affairs (2002). The Johannesburg Declaration on Sustainable Development. https://www.un.org/esa/sustdev/documents/WSSD_POI_PD/English/POI_PD.htm (accessed 10 January 2021).
- UNESCO (2019). Sustainable Development. https://en.unesco.org/themes/education-sustainable-development/what-is-esd/sd#:∼:text=Sustainable%20development%20is%20the%20overarching,to%20meet%20their%20own%20needs.%E2%80%9D (accessed 10 January 2021).
- Wahlen, C. B. (2019). UN report highlights environmental, health risks from E-waste, urges circular economy shift. https://sdg.iisd.org/news/un-report-highlights-environmental-health-risks-from-e-waste-urges-circular-economy-shift (accessed 15 March 2021).
- Yamane, L.H., Moraes, V.T., De Crocce, D. et al. (2011). Recycling of WEEE : characterization of spent printed circuit boards from mobile phones and computers. Waste Management 31 (12): 2553–2558. https://doi.org/10.1016/j.wasman.2011.07.006.
- Zeng, X., Gong, R., Chen, W.-Q., and Li, J. (2016). Uncovering the recycling potential of “new” WEEE in China. Environmental Science & Technology 50 (3): 1347–1358. https://doi.org/10.1021/acs.est.5b05446.
- Zhang, S., Ding, Y., Liu, B. et al. (2015). Challenges in legislation, recycling system and technical system of waste electrical and electronic equipment in China. Waste Management 45: 361–373. https://doi.org/10.1016/j.wasman.2015.05.015.
- Zhao, T., Ji, R., Yang, H. et al. (2019). Distinctive electrochemical performance of novel Fe-based Li-rich cathode material prepared by molten salt method for lithium-ion batteries. Journal of Energy Chemistry 33: 37–45. https://doi.org/10.1016/j.jechem.2018.08.005.
